Introduction
You have qualified your supplier, signed a quality contract, performed a supplier audit, and all ready to start working with the new and promising supplier. So, what’s next? How can you assure that the parts you receive are suitable for your needs?
When starting to work with a new vendor, which is also required to supply custom-made parts, a process should be performed to make sure the part is suitable, both for your production needs and in accordance with your quality demands. The PPAP (Purchased Part Approval Process) methodology can achieve these goals while concentrating on the approval process of a custom-made part from an approved supplier, all this while reinforcing the trust between you and your supplier.
PPAP definition and history
The PPAP process is defined as the approval process of new or revised parts or parts resulting from new or significantly revised production methods and or design changes. It is a valuable tool for establishing confidence in component suppliers and their production processes.
The PPAP process was initially developed in 1993 as part of quality assurance processes in the automotive industry by the Automotive Industry Action Group (AIAG). It is part of the five core quality tools in the Automotive industry:
- APQP (Advanced Product Quality Planning)
- PPAP (Production Part Approval)
- SPC (Statistical Process Control)
- MSA (Measurement System Analysis)
- FMEA (Failure Mode Effect Analysis)
Although PPAP is associated primarily with the automotive industry, similar agreements between component suppliers and purchasers have been adopted by other industries, including the Medical Device industry.
When to use PPAP
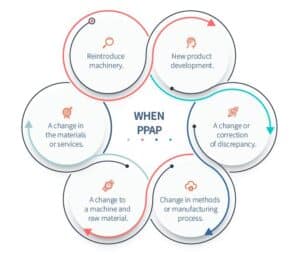
PPAP, as a method, can be used in a number of situations:
- New product development.
- A change or correction of discrepancy.
- Change in methods or manufacturing process.
- A change to a machine and raw material.
- A change in the materials or services.
- When the production tooling/machinery becomes inactive for production volume up to 12 months or more.
PPAP is not a process intended to approve suppliers, and it is not required for off-the-shelf parts.
The method comprises 18 sections intended to make sure the parts received from a supplier conform to the demands. Some of the parts included are- design documents, risk assessment documents, process flow diagrams, validation records (including test record validations, process validation, etc.), and other documents required for part approval.
Summary
Although the PPAP method is currently not mandatory, adopting it can assist in reaching regulation requirements set in international standards and regulations, such as ISO13485, MDR, and the FDA’s CFR 21 Part 820.
What we can suggest
RS NESS is a highly experienced service provider in the Medical industry.
We can assist you with a PPAP process implementation to help you create an appropriate and suitable process for your needs and improve your supplier quality.
Our expertise is in the field of Regulatory Affairs, Clinical Affairs, QA, Validation, and Engineering processes.
We differentiate ourselves by being quality-oriented. Our technical expertise comes with a hands-on experience and approach, ensuring that our clients receive the most effective and professional service.
Our clients range from small Start-Ups to international companies.
If you have any questions or need professional support, don’t hesitate to get in touch with us.
:About the author

Linor Skutelsky
QA Engineer and Project manager
Highly experienced in quality assurance in the Medical Device industry. In the past four years, Linor participated in many projects.
Linor earned her Bachelor of Science degree in Biotechnology Engineering from Ben Gurion University, Be’er Sheva, Israel, and her Masters of Science degree in Environmental Engineering from the school of Mechanical Engineering, Tel Aviv University, Tel Aviv, Israel.